
Previous studies demonstrated nMFS mutations tend to localize in an internal region of FBN1 called the neonatal region 10– 12. In contrast to classical MFS (cMFS), patients with the most severe form (neonatal MFS nMFS) rarely survive past the second year of life 8, 9. Connective tissue weakness caused by low-quality or decreased numbers of microfibrils is responsible for the primary clinical problems associated with MFS, including aortic aneurysm, dissection, and rupture 6, 7.Īlthough the natural history of MFS is weakened connective tissues, clinical presentations are highly variable. Fibrillin-1 forms the principal component of 10–12 nm diameter microfibrils, which are essential for elastic fiber assembly and structure 4, 5. The disorder is caused by mutations in FBN1, which encodes the calcium-binding extracellular matrix glycoprotein fibrillin-1 2, 3. Marfan syndrome (MFS) is an autosomal dominant connective tissue disorder that affects multiple body systems and organs 1. Such mechanosensitive “smart” features may represent novel mechanisms for mechanical hemostasis regulation in extracellular matrix that are pathologically activated in MFS. 
These results point to a potential mechanosensitive mechanism for fibrillin-1 in regulating extracellular transforming growth factor beta (TGFB) bioavailability and microfibril integrity. 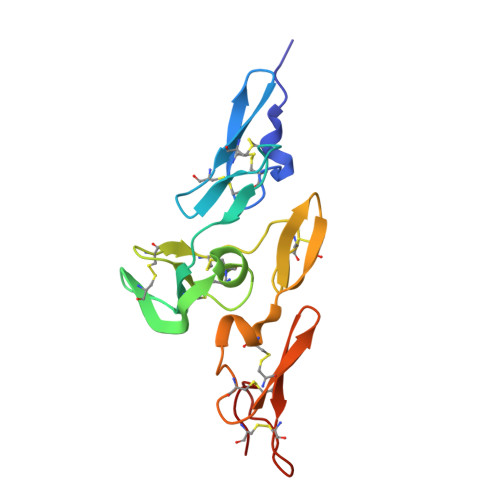
We further demonstrate the disulfide bonds in cbEGF domains uniquely orchestrate protein unfolding by showing that MFS disulfide bond mutations markedly disrupt normal mechanosensitive calcium binding dynamics. Using steered molecular dynamics (SMD) simulations, we demonstrate that cbEGF domain calcium binding decreases under mechanical stress (i.e. Perturbed calcium binding caused by cbEGF domain mutations is thus thought to be a central driver of MFS pathophysiology. Crucially, calcium plays a fundamental role in stabilizing cbEGF domains. This region is predominantly composed of eight calcium-binding epidermal growth factor-like (cbEGF) domains, each of which binds one calcium ion and is stabilized by three highly conserved disulfide bonds. Patients with the most severe form of MFS (neonatal MFS nMFS) tend to have mutations that cluster in an internal region of fibrillin-1 called the neonatal region. Marfan syndrome (MFS) is a highly variable genetic connective tissue disorder caused by mutations in the calcium binding extracellular matrix glycoprotein fibrillin-1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
